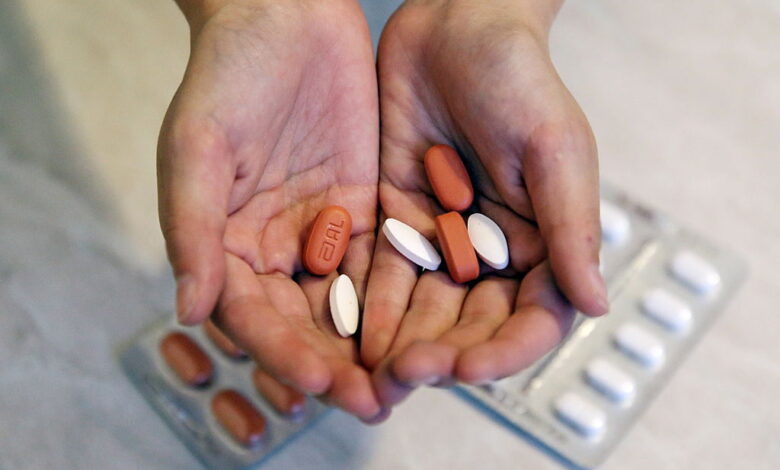
Moscow, Russia, November 26, 2016: A HIV-positive child's medicines. Photo Credit: Vyacheslav Prokofyev/Getty Images
Health SwitzerlandHIV Babies Now Have Access to Life-Saving Treatment
The World Health Organization (WHO) approved an HIV drug re-dosed and adapted for infants from one month old and onwards in order to give the best chance of survival to children afflicted with the disease.
WHO hails this as a “significant milestone with the potential to save many lives”. If babies receive the water-dissolvable and strawberry-flavored preparation of dolutegravir – one of the most important HIV drugs and the first choice for combating the virus – HIV is no longer detectable in 62% of them after six months of treatment.
The sooner children are treated, the better are their chances of winning their fight against the virus. Every year, some 160,000 boys and girls under 10 still live with HIV, and 90% of all underage patients come from sub-Saharan countries. Agreements have been made between the United Nations and manufacturers, so the drug will be affordable, even in the poorest regions of the world, where the preparation will be available at less than three euros a month.